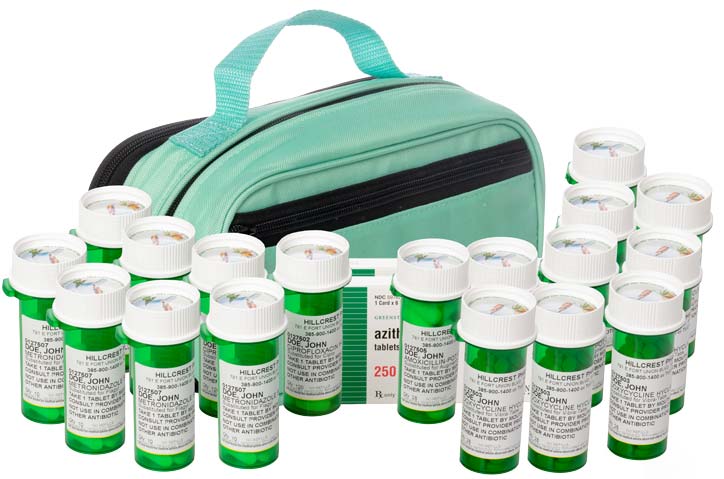
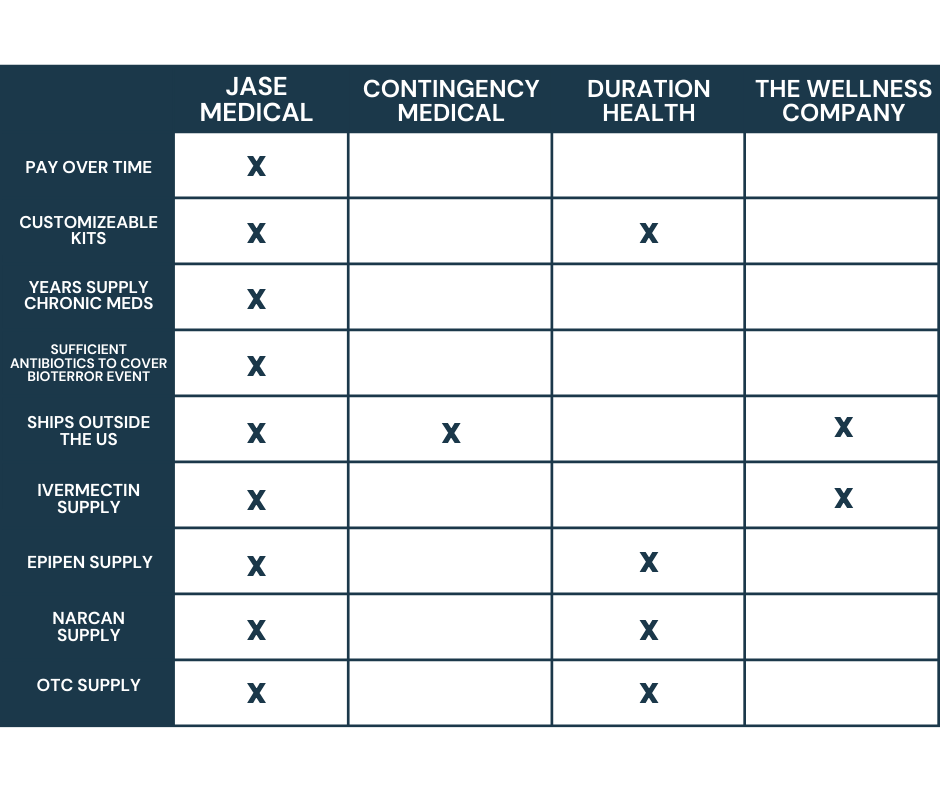
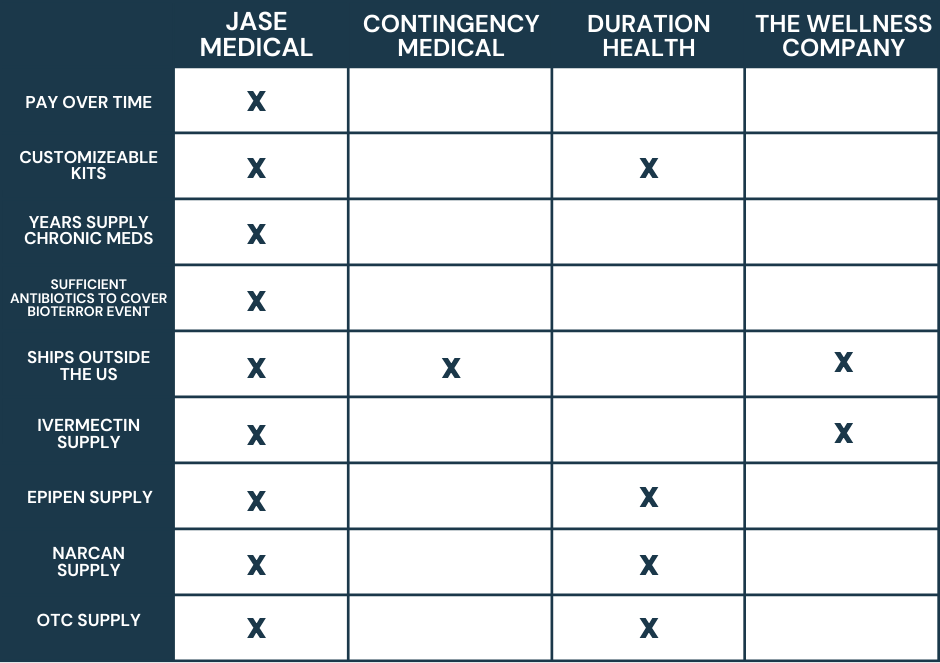
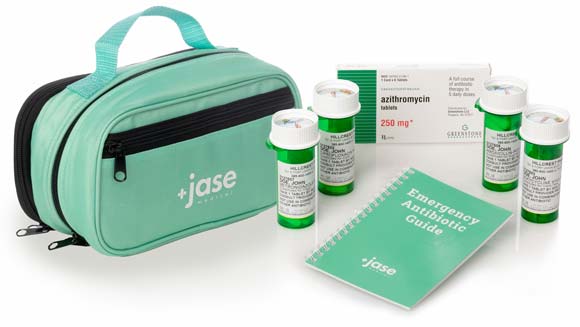
*By the Jase Medical Clinical team* Most Americans think of war as something that happens somewhere else, visible mainly through a screen. The Iran conflict has found its way into something far more intimate: the medicine bottles in your bathroom cabinet.Since Iran...
Food Poisoning: Causes, Prevention, and Treatment
With the right measures taken, this very common illness can also be very preventable.
With Father’s Day fast approaching and family meals being planned, food poisoning may be the last thing you want to think about. Following the food safety tips below will ensure everyone remembers your meal for the right reasons.
Food poisoning is a miserable experience that strikes millions each year, and many of us know it all too well. Caused by eating contaminated food or drinks tainted with harmful toxins, bacteria, viruses, or parasites, it’s mindlessly easy to get, but also largely preventable with the right precautions in place.
Read on to learn about what causes food poisoning, how to prevent it, and ways to treat it.
What makes us sick?
The culprits behind food poisoning are varied. Common bacterial offenders include salmonella, E. coli, and campylobacter. These can cause nasty cramps, nausea, vomiting, and diarrhea. Antibiotics may be prescribed for severe bacterial infections, but often rest and fluids are the best course of action.
Parasites like Giardia can also wreak havoc, causing similar symptoms along with bloating and gas. Anti-parasitic medication is typically needed to eradicate these unwelcome guests.
| According to the CDC: Each year 48 million people get sick from food poisoning, 128,000 are hospitalized, and 3,000 cases are fatal |
Prevention is Key
Follow these food safety tips and prevent getting sick in the first place!
- Safe Handling: Practice proper hygiene in the kitchen. Wash hands thoroughly before handling food, and clean surfaces regularly.
- Cook it Right: Ensure meats reach proper internal temperatures to kill bacteria. Use a food thermometer for accurate readings.
- Chilling Out: Refrigerate leftovers promptly and avoid reheating food multiple times.
- Beware BBQ’s and Buffets: Be cautious at buffets, potlucks and BBQ’s, especially during hot weather, as food can spoil more quickly.
Treatments for Food Poisoning
Treatment for food poisoning depends on the cause and severity of the symptoms:
- Bacterial Infections: Mild bacterial food poisoning often resolves without specific treatment. For severe cases, antibiotics like ciprofloxacin or azithromycin may be prescribed.
- Viral Infections: There is no specific treatment for viral food poisoning; supportive care, such as hydration and rest, is essential.
- Parasitic Infections: Antiparasitic medications, such as metronidazole or nitazoxanide, are used to treat parasitic infections.
In all cases, maintaining hydration is crucial, as food poisoning often causes significant fluid loss through vomiting and diarrhea.
Increased Risk in the Summer and During Travel
The risk of food poisoning is always present, but the warmer weather, or traveling to new destinations can increase the odds of encountering the dreaded belly rumble.
✈️ Food poisoning is more likely when traveling because your body is not used to the local bacteria in the food and water, potentially exposing you to pathogens that are not common back home. This is especially true when traveling to regions with different sanitation standards. Drinking bottled water, avoiding raw foods, and eating at reputable establishments can help reduce this risk.
☀️ Bacteria-based food poisoning is more prevalent in the summertime due to warmer temperatures, which promote bacterial growth. Foods left out at picnics, barbecues, or buffets can quickly become breeding grounds for bacteria – highlighted recently by the state of Oregon.
| This past week the FDA issued a warning, saying to take extra precautions while eating shellfish – particularly oysters and clams – after 21 people have been sickened in the northwest in the past month. |
Fortunately, most of the time passing the food, rehydrating, and resting are enough to recover. OTC medications like Loperamide (Immodium), and Bismuth subsalicylate (Pepto-Bismol) can help alleviate the symptoms during your recovery.
In more serious cases antibiotics such as Azithromycin, and Ciprofloxacin (both come in every Jase Case) can be used for food poisoning stemming from bacteria.
When it comes to food poisoning, be vigilant! Clean your cooking surfaces, wash your hands, and know your food sources! Always remember, when in doubt, throw it out. If food seems off, don’t eat it!
Lifesaving Medications
Everyone should be empowered to care for themselves and their loved ones during the unexpected.
Recent Posts
Keeping you informed and safe.
The War Nobody Prepared For: How the Iran Conflict Is Threatening Your Medicine Cabinet
Your Immune System Can Fight Cancer
*By the Jase Medical Clinical team* For decades, the dream in oncology has been simple and audacious: teach your own immune system to hunt cancer. Not poison it with chemotherapy. Not burn it with radiation. Train your body's existing defenses to do what they were...
The 2026 Tick Surge Is Already Here. And It’s Worse Than Doctors Predicted.
*By the Jase Medical Clinical team* I want to tell you something that caught my attention as a physician before it caught my attention as a CEO. A few weeks ago I was reading through case reports from colleagues in the Northeast. Family docs, internists, infectious...
Beyond Chemo: 10 Compounds Researchers Are Studying to Complement Cancer Care
Cancer treatment has always been a moving target. Chemotherapy, radiation, and surgery remain the pillars of oncology — but for decades, researchers have been quietly building a parallel body of literature around compounds that may support, enhance, or complement...